Hydrea 500 mg Capsules – Hydroxyurea Oral Antineoplastic & Sickle Cell Therapy
EGP3500
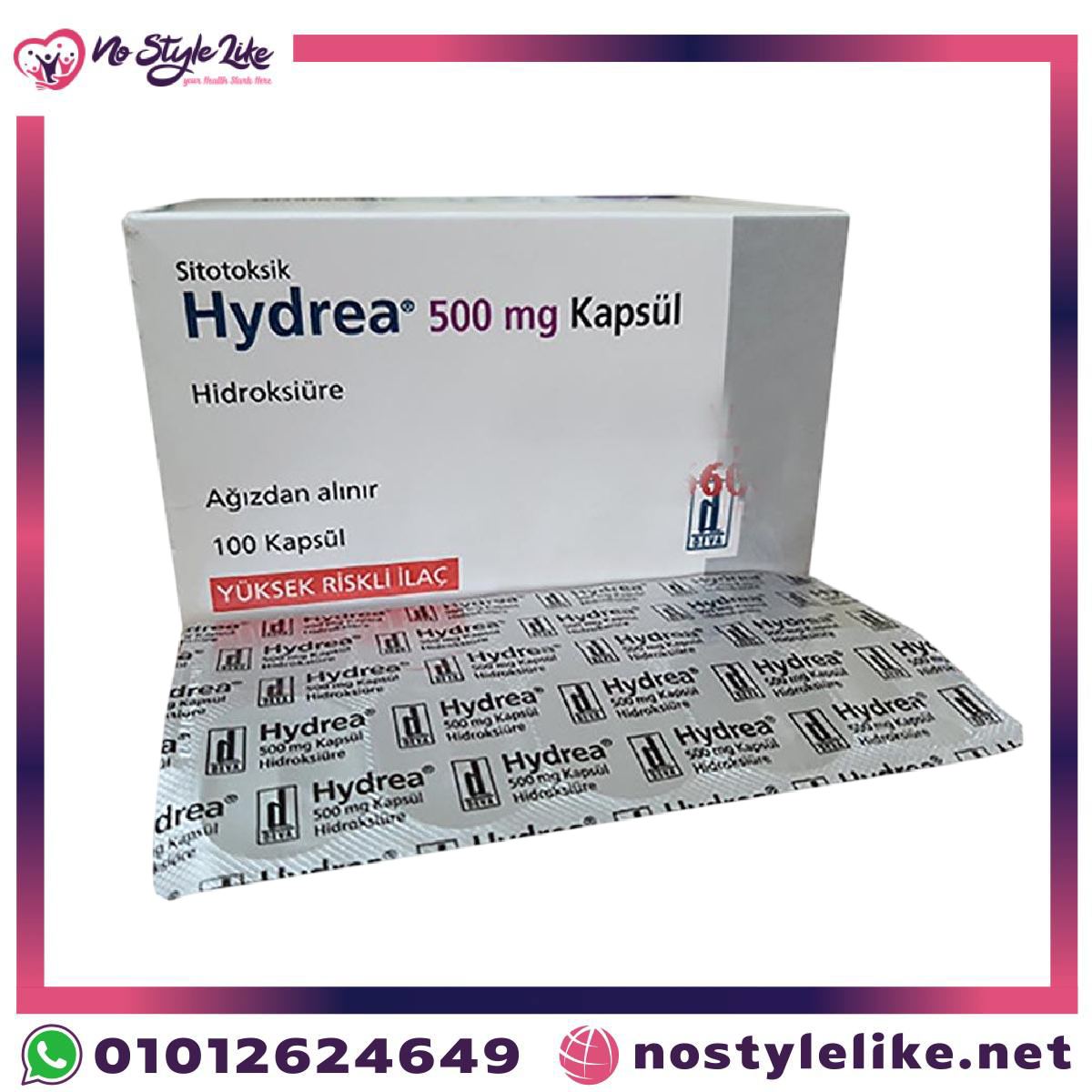
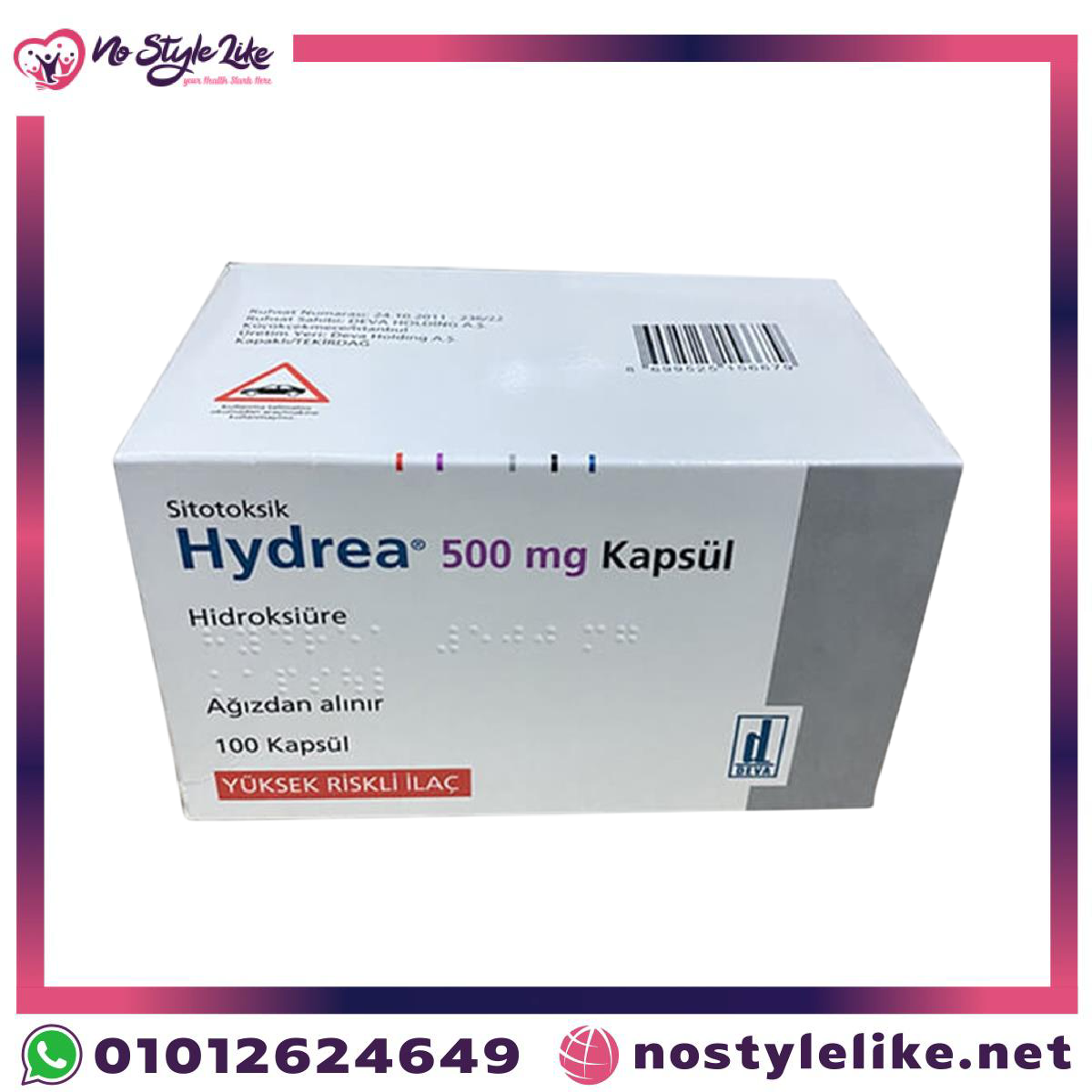
| Name | Hydrea 500 mg Kapsul |
|---|---|
| Brand | Bristol-Myers Squibb |
| Strength | 500 mg |
| Quantity / Size | 100 Capsules (per box) |
| Form | Capsules |
| Appearance | Opaque white hard gelatin capsules |
| Expiry Date | 8/2028 |
| Country of Origin | Turkey |
| Product Code | HYD500CAP |
1. Introduction to Hydrea 500 mg Capsules
Hydrea (hydroxyurea) is a locked prescription medication taken orally. It works by inhibiting DNA synthesis via ribonucleotide reductase blockade, slowing the proliferation of blood and cancer cells. Hydrea is an FDA-approved generic and essential medicine used widely since 1967.
2. Therapeutic Uses
Chronic Myelogenous Leukemia (CML): Hydrea helps reduce leukemic cell counts .
Sickle Cell Disease (SCD): Boosts fetal hemoglobin production, reducing painful vaso‑occlusive crises and transfusion needs in children and adults.
Head & Neck Cancer: Often combined with radiation for squamous cell carcinomas.
Other Myeloproliferative Disorders: Used in essential thrombocythemia, polycythemia vera, and occasionally psoriasis (Wikipedia).
Melanoma research: Emerging data suggests potential in certain melanomas via miRNA modulation .
3. Mechanism of Action
Hydrea inhibits ribonucleotide reductase, arresting cell cycle in G1/S phase and enhancing radiosensitivity. In SCD, it elevates fetal hemoglobin (HbF) to reduce sickling.
4. Dosage & Administration
Starting dose for adults with normal renal function: approx. 15 mg/kg once daily.
Renal impairment (CrCl <60 mL/min): dose reduced by 50% (≈ 7.5 mg/kg).
Pediatric care: Customized to body weight; tablets/capsules taken whole, with/without food .
Explore more: Natrol Acidophilus Probiotic 1 Billion (150 ct) – Digestive & Immune Support Supplement
5. Monitoring & Safety Precautions
Bone marrow suppression: Frequent CBCs before and during treatment to monitor WBCs, platelets, RBCs.
Infection risk: Watch for fevers, chills, sore throat .
Secondary malignancies: Rare skin cancer or leukemia risk with long-term use .
Pregnancy & fertility: Teratogenic; contraception recommended for 6 months post‑therapy in both genders.
Vaccinations: Avoid live vaccines while on therapy .
Sun protection: Use sunscreen; avoid excessive sun exposure.
6. Common & Serious Side Effects
| Side Effect Type | Examples |
|---|---|
| Common | Low blood counts, nausea, loss of appetite, headache, constipation, mouth sores |
| Serious | Fever/infections, bleeding/bruising, black/tarry stools, dyspnea, skin ulcers or gangrene |
If serious symptoms appear, stop Hydrea and seek medical attention immediately.
Explore more: Horbaach Vitamin D3 5000 IU + K2 MK-7 100 mcg | High Potency 180 Softgels
7. Handling & Storage
Handle with care; avoid contact if pregnant or via caregivers (use gloves).
Store at 15–30 °C, tightly sealed in original packaging, out of children’s reach.
8. Drug Interactions & Contraindications
Contraindicated: severe bone marrow depression, prior hypersensitivity, severe anemia, during pregnancy.
Interactions: other myelosuppressive agents or recent radiation increase marrow toxicity risk; adjust dose accordingly.
9. Why Choose Hydrea?
Well‑studied with 50+ years of use.
Generic & essential medicine – high cost‑effectiveness.
Versatile indications include sickle cell, CML, head/neck cancers, etc.
✅ FAQs
Q1: How long does it take for Hydrea to reduce sickle‑cell crises?
A: Most patients observe fewer painful crises and transfusion needs within 2–4 months of consistent dosing.
Q2: Can Hydrea be used during pregnancy?
A: No—Hydrea is teratogenic. Effective contraception is required during treatment and for 6 months after therapy ends.
Q3: What types of blood monitoring are needed?
A: Complete blood counts (CBC) are done weekly during initiation, then monthly once stabilized to watch for cytopenias .
Q4: Can Hydrea cause hair loss or skin ulcers?
A: Yes. Hair thinning, nail discoloration, and rare skin ulcers/gangrene have been reported—contact your physician if skin lesions occur.
Q5: Could Hydrea increase the risk of other cancers?
A: Long-term use in myeloproliferative disorders has a small risk of secondary leukemia or skin cancer—regular skin checks are advised.
Explore more at No style Like : Cancer treatment drugs
User Reviews
⭐⭐⭐⭐⭐ Very effective for SCD “After starting Hydrea, my child hasn’t had a pain crisis in over four months—life‑changing!”
⭐⭐⭐⭐ Good for leukemia but tough monitoring “It helps control my CML, but I do worry about frequent blood tests.”
⭐☆☆☆☆ Caused mouth ulcers “I developed painful mouth sores and had to pause treatment.”